 Similar processes are often used in sequence – for example, a barium nitrate solution will react with sulfate ions to form a solid barium sulfate precipitate, indicating that it is likely that sulfate ions are present. To identify the cation, the color of the precipitate and its solubility in excess are noted. To do this, an alkali first reacts with the unknown salt to produce a precipitate that is the hydroxide of the unknown salt. Precipitate formation is useful in the detection of the type of cation in a salt. Inorganic chemistry Precipitation in aqueous solution If energy changes are not favorable, or without suitable nucleation sites, no precipitation occurs and the solution remain supersaturated. This involves energy changes depending on the dissolution reaction free energy ( endothermic or exothermic process accompanied by an entropy increase) and the relative surface energy developed between the solid and the solution. The creation of a solid particle implies the formation of an interface with the solution. When a potassium iodide solution reacts with a lead(II) nitrate solution, a yellow precipitate of lead(II) iodide is formed.Īn important stage of the precipitation process is the onset of nucleation. When a barium chloride solution reacts with sulphuric acid, a white precipitate of barium sulfate is formed. The formation of a precipitate can be caused by a chemical reaction. Precipitation occurs more rapidly from a strongly supersaturated solution. This can be due to temperature changes, solvent evaporation, or by mixing solvents. 
The precipitation of a compound may occur when its concentration exceeds its solubility. The notion of precipitation can also be extended to other domains of chemistry (organic chemistry and biochemistry) and even be applied to the solid phases ( e.g., metallurgy and alloys) when solid impurities segregate from a solid phase. The clear liquid remaining above the precipitated or the centrifuged solid phase is also called the ' supernate' or ' supernatant'. In case of an inorganic chemical reaction leading to precipitation, the chemical reagent causing the solid to form is called the precipitant. 
The solid formed is called the precipitate. In an aqueous solution, precipitation is the process of transforming a dissolved substance into an insoluble solid from a super-saturated solution. Trying to ride it out alone can make an already challenging experience even more difficult.Principle of chemical precipitation in aqueous solution 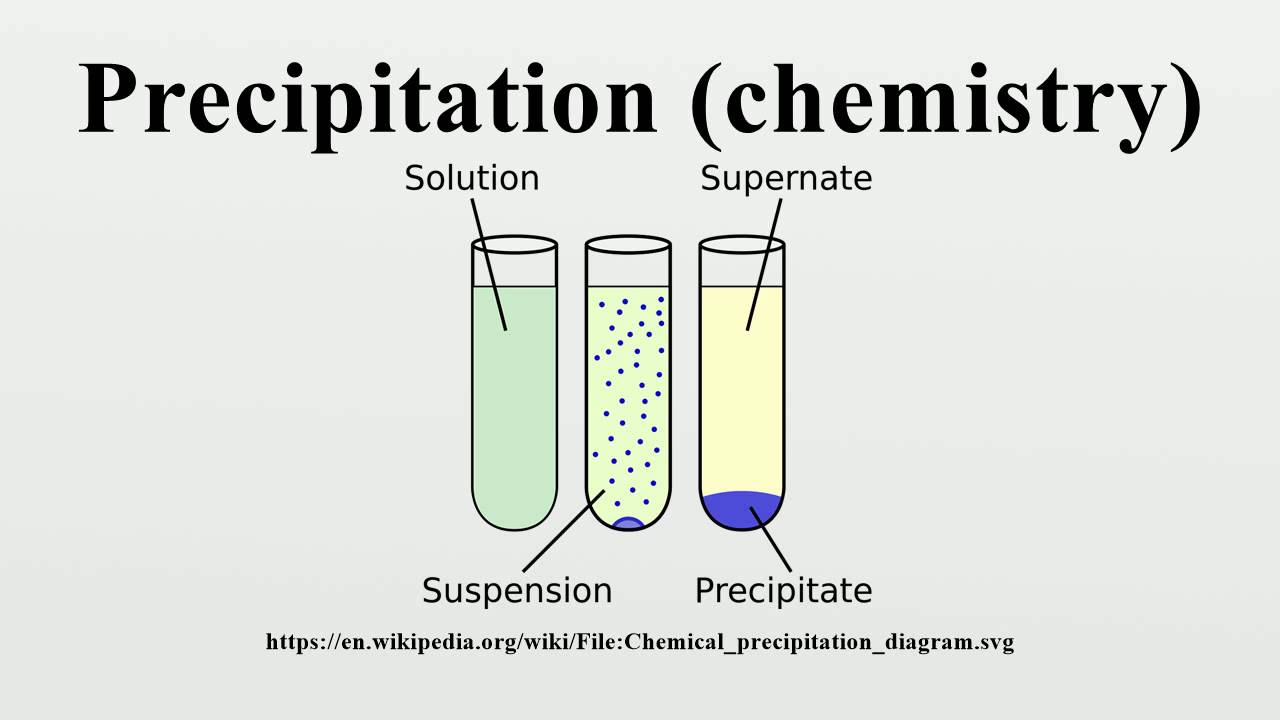
Get medical help: If you begin to experience precipitated withdrawal symptoms, contact your healthcare provider as soon as possible.If you are in pain, you can take over-the-counter painkillers like paracetamol. If you have diarrhea, you can take drugs like Imodium. Treat symptoms as they occur: With your healthcare provider's recommendations, you can treat symptoms as they crop up.Reduce stress: Eliminating stressors within your control can help you stay relaxed and focused on recovering from your symptoms.You can alternate between drinking water and drinks with electrolytes and sucking on ice cubes or eating foods with high water content, such as watermelons. Understandably, doing anything, including drinking water regularly, can be difficult. All of these can cause you to become dehydrated very quickly. It’s common for excessive sweating, diarrhea, and vomiting to occur. Drink a lot of water: Staying well hydrated is crucial when experiencing precipitated withdrawal. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
